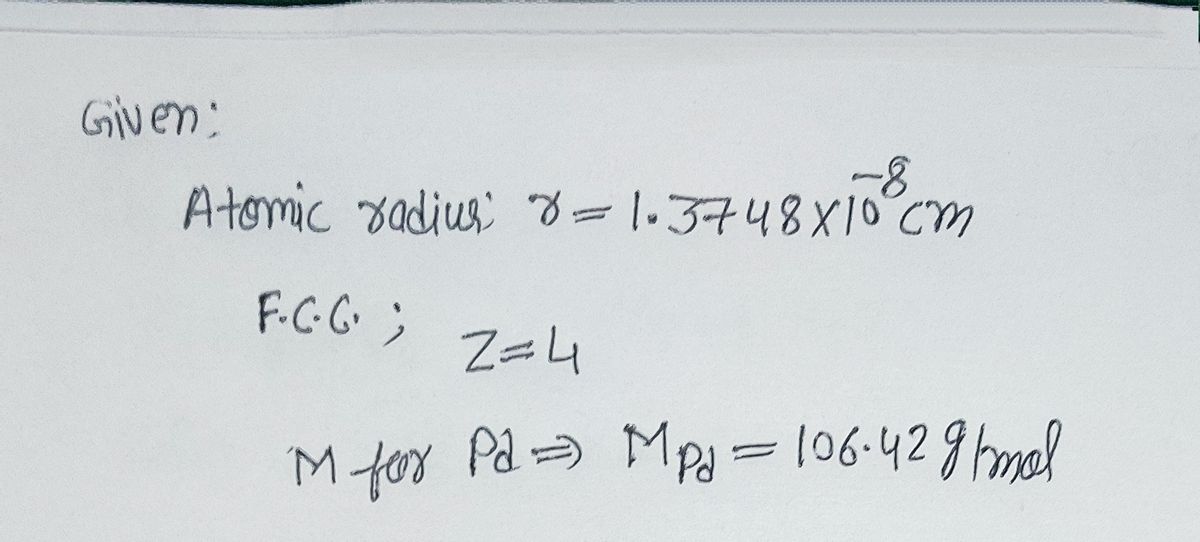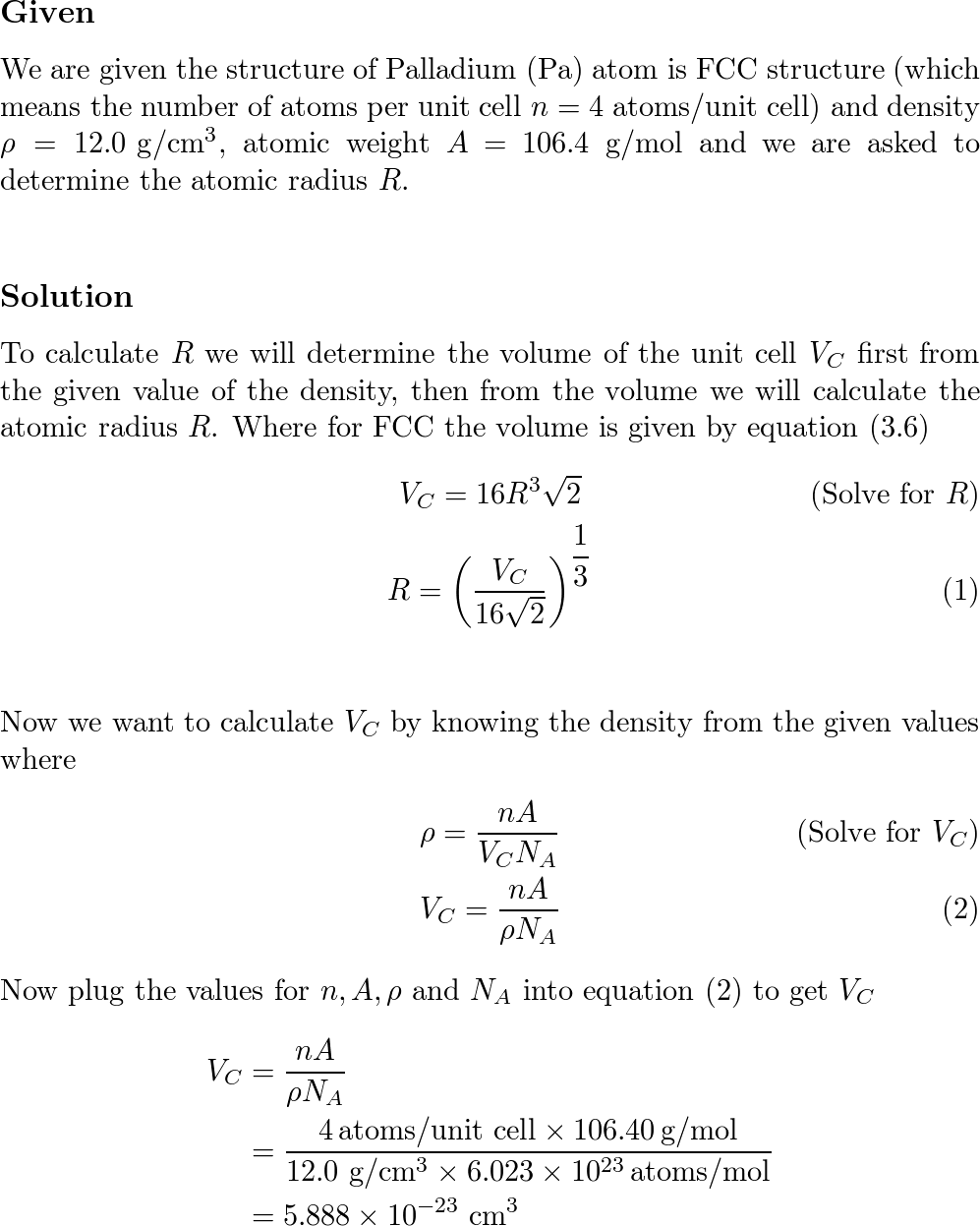a) Density of state of Pd-Rh-H system at ambient pressure. (b) Density... | Download Scientific Diagram

Density functional study on the mechanism for the highly active palladium monolayer supported on titanium carbide for the oxygen reduction reaction: The Journal of Chemical Physics: Vol 144, No 20

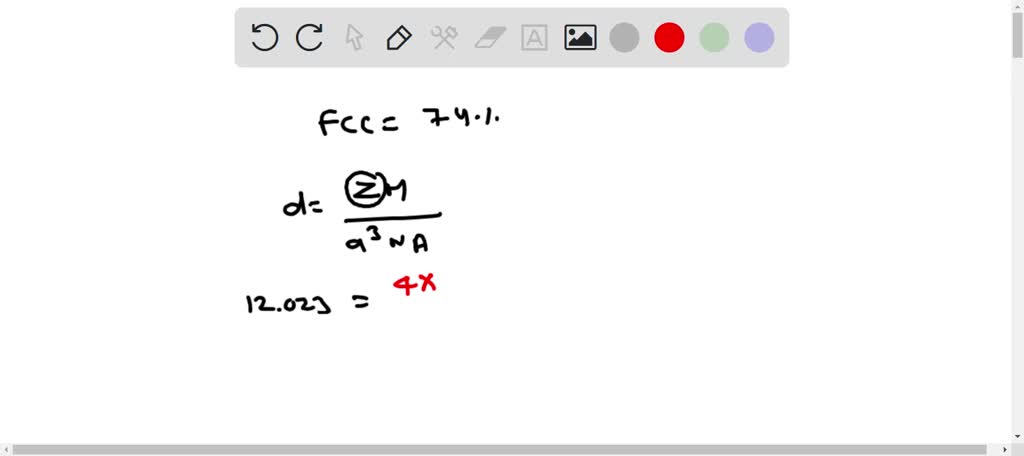
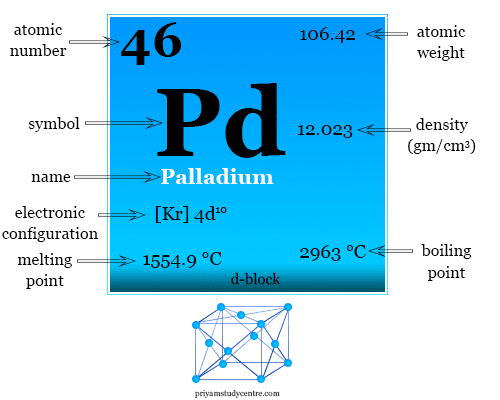
The atomic radius of palladium is 1.375 A. The unit cell of palladium is a face-centred cube. Calculate the density of palladium.

The atomic radius of palladium is 1.375 A. The unit cell of palladium is a face-centred cube. Ca... - YouTube

SOLVED:If palladium metal (density 12.0 g / cm^3 ) dissolves 935 times its own volume of H2 at STP , what is the value of x in the formula PdHx ? What

Electron density for the bulk of palladium and one hydrogen atom in the... | Download Scientific Diagram

0.39" Element Cube Palladium 10mm Density Cubes for Periodic Table Collection High Purity Element Collections (0.39", Palladium)| | - AliExpress

Palladium Pd Metal 10mm Density Cube 99.95% Pure For Element Collection - Magnetic Materials - AliExpress

the density of palladium is 12.0g/Cm^3. what volume in liters would be occupied by 532 g of - Brainly.com

Lattice-strain and electron-density modulation of palladium nanocatalysts for highly efficient oxygen reduction - ScienceDirect

the density of palladium is 12.0g/Cm^3. what volume in liters would be occupied by 532 g of - Brainly.com